The Nobel Prize in Physiology or Medicine 2025 was awarded to Mary E. Brunkow, Fred J. Ramsdell, and Shimon Sakaguchi for their discoveries concerning peripheral immune tolerance — a breakthrough that reveals how our immune system prevents itself from attacking the body it was built to defend.
(Source: NobelPrize.org)
Interestingly, this discovery mirrors another remarkable Nobel-winning insight from 2016, when Yoshinori Ohsumi was honored for discovering autophagy, the process by which cells eat and recycle themselves to stay healthy.
Together, these two Nobel Prizes tell a poetic story of balance through self-regulation — one at the level of a single cell, the other at the level of the entire immune system.
2025 Nobel Prize in Medicine — How the Immune System Learns Not to Attack Itself
What problem did it solve?
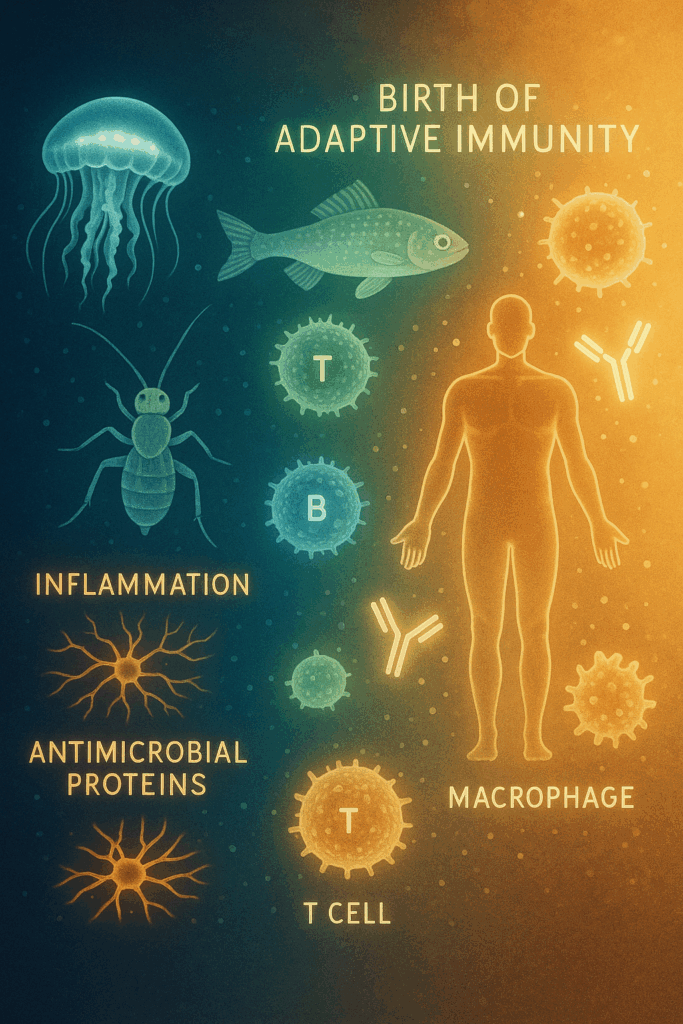
Our body’s immune system is an army built to defend us against invaders such as viruses, bacteria, and cancer cells. But this same army can become dangerous if it turns its weapons inward, mistaking our own cells as enemies. When this happens, we suffer from autoimmune diseases like type 1 diabetes, lupus, or rheumatoid arthritis.
Scientists already understood part of how this control worked through a process called central tolerance, where the immune system eliminates overly aggressive cells during early development. However, this could not explain how immune balance was maintained throughout life in the body’s tissues — what scientists call the periphery.
This is where the 2025 laureates made their landmark contribution.
What did they discover?
They identified the regulatory T cells (T-regs) and the Foxp3 gene, which act as the master controllers of immune tolerance.
- Regulatory T cells — the peacekeepers
- Discovered by Shimon Sakaguchi in the 1990s, these T-regs act as the brakes of the immune system.
- Their role is not to attack pathogens but to restrain other immune cells from attacking the body itself.
- They maintain peace and prevent autoimmune chaos.
- The Foxp3 gene — the control switch
- In 2001, Mary E. Brunkow and Fred J. Ramsdell discovered that mutations in the Foxp3 gene lead to fatal autoimmune disorders in both mice and humans.
- This gene is essential for the development and proper function of regulatory T cells.
- When Foxp3 is faulty, the immune system loses its “braking system,” leading to diseases like IPEX syndrome, where the body attacks multiple organs.
By connecting T-regs with Foxp3, these scientists revealed how the immune system achieves peripheral immune tolerance — a form of ongoing self-control that keeps our internal peace intact.
Why this matters for medicine
This discovery opens vast new doors for treatment and research:
- Autoimmune diseases: Enhancing or restoring T-regs could calm harmful immune attacks.
- Organ transplantation: Boosting tolerance could help the body accept donor organs.
- Cancer therapy: Adjusting T-reg activity can help the immune system better target tumors.
- Drug safety: Understanding immune restraint helps minimize unwanted inflammatory reactions.
Their research lays the foundation for an entirely new field of immune regulation therapies — where medicine learns from nature’s own systems of balance and self-restraint.
A simple analogy
Imagine your body as a high-security building:
- Guards = immune cells that patrol for intruders.
- Supervisors = regulatory T cells that stop guards from mistakenly arresting the staff.
- Foxp3 = the instruction manual that trains these supervisors.
Without Foxp3, the supervisors vanish, and the guards start attacking everyone — even their own building.
2016 Nobel Prize in Medicine — The Cell That Eats Itself to Survive
Winner: Yoshinori Ohsumi (Japan)
Discovery: The mechanism of autophagy — how cells “self-eat” to recycle their components.
What is autophagy?
The word autophagy comes from Greek: auto (self) + phagy (to eat).
It’s a process through which our cells clean up and renew themselves by breaking down and recycling old or damaged parts.
- When cellular components like proteins or organelles wear out, the cell wraps them in a membrane and digests them.
- The useful molecules are reused to build new structures.
- It’s like a self-cleaning oven or recycling plant inside each cell.
Why is it vital?
Autophagy keeps cells healthy, youthful, and adaptable:
- It prevents the buildup of toxic waste linked to diseases like Alzheimer’s and Parkinson’s.
- It helps cells survive stress and starvation by generating new energy.
- It even helps fight infections by digesting invading microbes or damaged regions.
In essence, autophagy is controlled self-destruction for the sake of renewal — the cell sacrifices its parts to preserve its wholeness.
The Connection: Two Halves of the Same Wisdom
The 2016 and 2025 Nobel Prizes appear to describe opposite phenomena — one about destruction, the other about restraint. Yet, together they express nature’s grand philosophy of balance.
| Process | Purpose | Type of Balance |
|---|
| Autophagy (2016) | Recycles damaged cellular components | Inner balance within one cell |
| Peripheral Immune Tolerance (2025) | Prevents immune cells from attacking the body | Systemic balance between immune cells |
Both mechanisms show self-regulation as the foundation of life:
- Autophagy teaches that to live, one must know what to let go of.
- Immune tolerance teaches that to protect, one must know when to stop fighting.
One cleanses by controlled destruction, the other preserves by controlled restraint.
One maintains order inside the cell, the other maintains peace across the body.
Together, they reveal that health is not the absence of conflict, but the mastery of control — a harmony between renewal and restraint.